Intentional and unintentional introduction of invasive and non-native species
Non-native species are those that do not naturally occur in an ecosystem. A non-native species is considered invasive when it is capable of aggressively establishing itself and causing environmental damage to an ecosystem. Plants, animals, and pathogens all can be invasive.

Typical traits of an invasive species include: 1) generalist; being able to survive in a variety of physical and biological situations, 2) rapid reproduction, growth, and dispersal ability, and 3) lacking natural predators or pests in the invaded ecosystem. Thus, invasive non-native species are successful competitors in new ecosystems, usually displacing native species and disrupting ecosystem processes. An increase in invasive non-native species is associated with land cover change (human development and seral stage) and habitat fragmentation, human activities that transport the plants and animals or their eggs/seeds, and to changes in disturbance regimes (Hobbs 2000).
Invasive non-native species are a worldwide problem; in the United States alone an estimated 50,000 non-native species have either been introduced or escaped within natural or managed ecosystems (Pimentel et al 2000). With that many species involved, the fraction that is invasive does not have to be large to inflict great harm upon native species and natural ecosystems. For example, 602 of the 1055 native plant species and 68 out of 98 native bird species that are categorized as threatened in the United States are imperiled by invasive non-native species (Gurevitch and Padilla 2004).
Invasive non-native species are either introduced intentionally, with the express purpose being the translocation of the organism or unintentionally as a secondary byproduct (Ruiz and Carlton 2003). A few examples of unintentional introduction include: ballast water exchange, packing material, and pathogens hitchhiking on other organisms. Identifying pathways and vectors is critical because the easiest means to prevent and reduce the spread of new invasions is vector interception or disruption (Carlton and Ruiz 2005). Without managing the pathways and vectors by which invasive non-native species enter the Salish Sea ecosystem, the number of successful establishments of invasive non-native species will increase.
Placeholder: Economic Consequences
Pressure: Invasive and Non-native Species in Salish Sea Ecosystem
In the following section, we use DPSIR terminology to help evaluate invasive species as a pressure to the ecosystem in terms of impacts to native populations and communities with intentional and unintentional introductions as drivers (Figure 5). The strategies to control and prevent invasive species are discussed in more detail in Chapter 4.
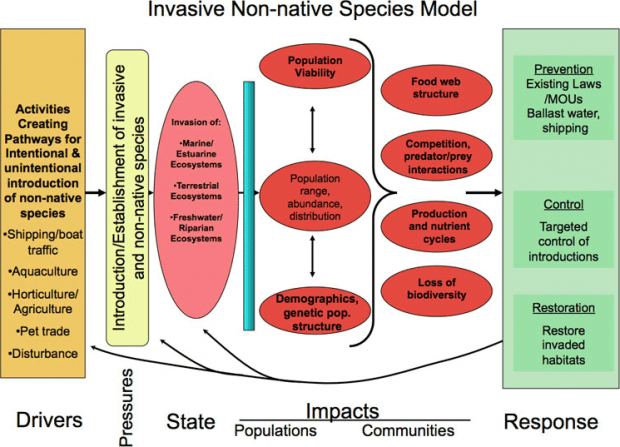
Figure 6. Driver-Pressure-State-Impacts-Response conceptual model for Invasive Species.
The Washington Invasive Species Council has identified approximately 700 invasive non-native species that have been introduced/established in and around Washington State at varying spatial extents (Washington Invasive Species Council 2009). Of these 700 species, the council identified 50 priority species/guilds based on these having highest impacts to the system. Of the top 50 priority species/guilds, 37 already occur in Washington and consist of 17 terrestrial plants, 2 terrestrial animals, 8 aquatic plants, 7 aquatic animals, and 3 insects/diseases. To give an idea as to the breadth of species considered in the top 50 invasive non-native species, a few examples include the following: knotweeds, butterfly bush, feral swine, spartina, caulerpa seaweed, New Zealand mud snail, tunicates, nutria, wood boring beetles, and viral hemorrhagic septicemia virus.
State: Invasion of Terrestrial Ecosystems
Clear differences have been demonstrated between invasive and non-invasive plant species on the basis of physiology, leaf-area allocation, shoot allocation, growth rate, size and fitness (van Kleunen et al 2010). Currently, the 2010 Washington State Noxious Weed list identifies 112 invasive non-native terrestrial plant species occurring throughout Washington . These species are classified into three major classes:
- Class A- Composed of 28 invasive non-native species (5 are considered in the 50 priority species/guilds) with limited distribution in Washington and eradication is required by law.
- Class B- Composed of 55 invasive non-native species (11 are considered in the 50 top priority species/guilds), which are presently established in limited portions of the state, with containment as the primary goal.
- Class C- Composed of 29 invasive non-native species (1 is considered in the 50 top priority species/guilds) that are widespread in Washington, with flexibility of control at the local level.
For terrestrial animals there is no comparable comprehensive list of species present throughout the Puget Sound region like there is for aquatic environments. However, some individual counties have partial lists of non-prioritized invasive non-native species that are present. For example, King County identifies the European Starling, House Sparrow, Eastern Gray Squirrel and domestic cat as invasive within the county .
Impacts
Two of the most influential factors influencing land invasions are disturbance and the transport of species by global trade. The facilitation between land transformation and global transport of species is bi-directionally linked. Land transformation provides opportunities for invasion and invasions can enhance and drive land transformation (Hobbs 2000). When both of these pressures interact, they create the potential for extreme changes of ecosystem dynamics (Hobbs 2000).
Fragmented native vegetation adjacent to human development is more likely to be invaded because of its interface with anthropogenic vegetation (edges) that enhances the spread of invasive species from the disturbed edges. The interaction between fragmentation and invasion results in changes in ecological processes, loss of native species and an overall reduction in biological diversity (Murcia 1995; With 2002). There is a coupling between disturbance (both natural and anthropogenic) and high levels of invasive non-native species. For example, Seattle public lands are highly disturbed by urbanization and also contain a large proportion of invasives. From 1999 to 2000, a citywide survey of Seattle’s 3,215 hectares of public land was completed. The survey found that invasive non-native plant species are present in 94% of these urban natural areas and that 20% of the city’s forested areas are highly invaded (Ramsay et al 2004). A follow-up sample performed in 2005 within the city’s eight forest types, indicated that these lands were still highly invaded (Seattle Urban Nature Project 2006). In the mixed coniferous/deciduous forest type for example, the regenerating tree layer is composed of 55% non-native evergreen trees and 8% non-native deciduous trees. Overall biodiversity and ecosystem function typically become reduced when invasive species become dominant (Sanders et al 2003). With invasive non-native species composing more than half of all regenerating trees found, the forest is more susceptible to greater damage from disease, pests and other disturbances thereby jeopardizing the future of this forest type (Seattle Urban Nature Project 2006).
Farther from urban centers, clear-cut logging is a major source of disturbance in the Cascades (Parks et al 2005). Throughout the Coast Range and Western Cascades, invasives dominate for the first 2-5 years following the disturbance but are then replaced by native species as succession progresses. This pattern was observed from 2001-2005 across the Forest Inventory and Analysis Program plots in Washington and Oregon mountain ranges (Harrington et al 2007). They found that the percentage of invasive species declined with increasing stand size class. Since larger stand class size is highly associated with time since a major disturbance, most invasive plant species on forest land in the region are associated with recently-disturbed locations.
However, even though native plant species regain dominance over invasives after a forest disturbance, there are long term effects on the dominant native species (Dale and Adams 2003). During the first 10 years following the debris avalanche of Mount St. Helens, plots inundated with invasive species had significantly greater conifer sapling mortality and lower native species diversity than un-invaded plots. After this initial 10 year period, no difference in conifer mortality was noted and native species diversity was higher within invaded plots. Even so, the plots dominated by invasive species still had fewer conifer trees overall. Thus, the short-term conifer mortality increase associated with non-native species invasion appears to have long-term effects on the recovery of conifers as the dominant vegetation. The reduction of these foundational conifer species may cause cascading effects, affecting energy and nutrient fluxes, hydrology, biodiversity and food webs (Ellison et al 2005).
Global travel and commerce has redistributed species around the globe, connecting regions that historically were biogeographic barriers. A nation’s non-native species diversity is strongly related to its level of trade (Westphal et al. 2008) and the United States is one of the leading nations in recipients of non-native invasives through international trade (Jenkins and Mooney 2006). While air- and seaports are major entry points for international trade, the commodities arriving at these destinations and those arriving from interstate commerce, are subsequently moved by road and rail. Many of these shipments contain unintended stowaways, such as untreated wood harboring non-native invasive insects and pathogens (Piel et al 2008). The travel corridors then help direct the movement of non-native invasives through less hospitable habitat, facilitating their spread and establishment (Hulme 2009).
Urban areas in the Puget Sound region are at high risk for introduction of non-native invasive bark- and wood-infesting insects based on the amount of urban and exurban forestland and the tonnage of imported goods (Colunga-Garcia et al 2009). The most prevalent pathway is imported machinery and nonmetallic mineral products originating from Asia (Colunga-Garcia et al 2009). There have been several incidences of Asian and citrus long-horned beetles being found in warehouse and plant nursery shipments to Washington State. Some beetles escaped into neighboring greenbelts, necessitating the cutting of several thousand trees, injections of a systemic pesticide, and the quarantine of all host material for a one-half mile radius around the beetle introduction site4.
Other non-native invasives are spread intentionally for human use, for example agriculture, horticulture or pet trade. The majority of woody non-native invasive plants in the United States were introduced for horticultural purposes—82% of 235 woody plant species identified as colonizing outside of cultivation have been used in landscaping (Reichard and White 2001). A conservative number of 104 non-native invasive shrub species are known in the United States, with at least 17 species occurring in Washington State (Boyce 2009). Many of these shrubs affect native forests by crowding out native species, reducing biodiversity and may change ecosystem functioning effectively halting successful tree regeneration (Boyce 2009). Shrubs are often introduced by escaping from gardens, where they are grown for their flowers and fruit. Birds and mammals are responsible for furthering the spread of the shrubs due to the large fruit crops produced. Birds alone are vectors for the seed dispersal of over 70 non-native shrub species nationwide (Boyce 2009).
State: Invasion of Marine/Estuarine Ecosystems
Coastal estuarine and marine ecosystems are among the most heavily invaded systems in the world (Grosholz 2002), mostly due to intentional and unintentional introductions by boat traffic (ballast water and hulls), aquaculture, bait and released pets (Carlton 2000).
Even though estuarine and marine systems are heavily invaded, currently, the 2010 Washington State Noxious Weed list identifies only 4 invasive non-native estuarine/marine plant species occurring throughout Puget Sound . However, all 4 are considered Class A species meaning eradication is required by law.
The Washington Department of Fish and Wildlife maintains a watch list of aquatic nuisance species of Washington’s marine and freshwaters. These non-native species are considered to have a high risk of becoming invasive and are separated into species of primary concern—those considered to have the highest level of environmental risk, and secondary concern—considered to have a lower level of environmental risk. According to the 2008 watch list, of the marine animals, 8 of 9 primary and 22 of 34 secondary species of concern are currently present in Washington. All but two of the primary marine species of concern overlaps with the top 50 priority species/guilds listed by the Washington Invasive Species Council. While a total of 4 marine secondary species of concern overlaps with the top 50 priority species.
Impacts
A study by Lawrence and Cordell (2010) looked at how ballast water influences the amount of propagules (e.g., larvae) of non-native species found in Puget Sound waters. Cordell’s results indicate that the Puget Sound receives an annual average of 7.5 x 106 m3 of ballast water from both foreign (mostly trans-pacific) and domestic waters. Foreign trans-Pacific vessels carried significantly fewer (p < 0.001) propagules compared to ships on domestic west coast routes. Of the propagules detected, trans-Pacific ships contained almost twice as many non-native species (19 species) than did those from ships on west coast routes (10 species), with seven species being common to both. However, even though trans-Pacific vessels had higher diversity of non-native species, densities of nonnatives were 100-200% greater in domestic ballast water. Considering that a variety of biological and physical factors affect an invader’s success, both foreign (high diversity) and domestic (high density) sources of ballast water have high potential to result in successful invasions of the Sound.
Wonham and Carlton (2005) reviewed the literature documenting 123 introduced invertebrate, algal, fish and vascular plant species in the Northeastern Pacific Ocean. They found the major invasion pathways to be shipping (hull fouling, solid and water ballast) and shellfish (particularly oysters) and finfish imports. Successful invasions increased at linear, quadratic, and expotential rates for different taxa, pathways and regions in the Northeastern Pacific. Of the regions included in this study, Puget Sound had the most introduced species.
Ballast water is not the only vector for distributing invasive non-native species in the Sound. Of the 62 established invertebrate invasive non-native taxa found in Puget Sound waters, only 25 are spread by ballast (Simkanin et al 2009). Six of these 25 taxa are exclusively distributed by ballast. Other major sources include ship fouling (35 taxa) and commercial oysters (39 taxa) (Simkanin et al 2009). Sixteen and 17 taxa are distributed exclusively by fouling and commercial oysters respectively.
Regardless of invasion pathway, invasive non-native marine/estuarine species in the Puget Sound are capable of causing extensive ecological changes. For example, highly invasive non-native cordgrass (S. alterniflora, S. anglica, S. patens, and S. densiflora) in estuarine habitat rapidly converts bare mudflat into a cordgrass monoculture. S. alterniflora was accidentally introduced in the 1890s when it was used as packing material for oysters shipped from the Atlantic coast (Grevstad et al 2003), it is most widely spread in Willapa Bay, infesting approximately 465 solid hectares (Phillips et al 2008). S. anglica was introduced in Port Susan Bay in 1961 for erosion control and cattle forage and infested approximately 36 solid hectares (Phillips et al 2008). S. patens and S. densiflora introduction pathways are unknown and take up less than 0.40 solid hectare at the mouth of the Dosewallips river and Gray’s Harbor (Phillips et al 2008). These infestations of Spartina have negative community level effects as it greatly reduces habitat available for fish, shellfish (commercial and native), migratory waterfowl and shorebirds (Hacker et al 2001; Buchanan 2003; Grevstad et al 2003; Semmens 2008).
When Spartina invades a variety of potential niches, physical conditions of the habitat are the main limiting factors controlling the high variation in establishment and growth among habitats rather than biological interactions (Dethier and Hacker 2005). Thus, the range, abundance and physical and biological effects of Spartina do vary depending on the type of habitat invaded (Hacker and Dethier 2006). Of four habitat types considered (mudflat, cobble beach, low and high salinity marsh), Spartina has the greatest range and abundance in mudflats and low salinity marshes compared to high salinity marshes and cobble beaches. Changes in sediment characteristics also substantially differed among habitats; some habitats experience greater accretion (mudflats), greater water content (cobble beach), and greater salinity loss (high salinity) than other habitats. Finally, native plant diversity declined in low salinity marshes but either increased or remained stable within the other habitat types, although percent cover and species richness of native macroalgae decreased. Thus, if changes occur in salinity, sea level, or sediment supply in various invaded habitats, Spartina impacts will be altered, most likely to the detriment of the native community (Hacker and Dethier 2006).
Another estuarine invasive non-native ecological engineer, an eelgrass (Z. japonica), has had an opposite community effect. Eelgrass beds provide habitat and food to a wide variety of marine organisms, protection for fry, and prevent beach erosion, thus being a critical component of the nearshore ecosystem. The invasive form of eelgrass typically does not coexist or compete with the native eelgrass (Z. marina) of Puget Sound but simply extends the eelgrass bed further into the upper intertidal zone (Britton-Simmons et al 2010). Within two decades of introduction, Z. japonica almost doubled the total eelgrass habitat in Boundary Bay, British Columbia (Williams 2007). Now migrating waterfowl prefer it over native eelgrass as their principal food. Z. japonica has increased faunal diversity, net primary production and influenced the biogeochemistry of the entire estuary (Williams 2007).
These two invasive non-native species, S. alterniflora and Z. japonica also have indirect effects on each other (Williams 2007). The vector of the interaction depends on which colonizes first. If S. alterniflora colonizes first, it outcompetes Z. japonica. However, if Z. japonica colonizes first it inhibits the seed germination of S. alterniflora.
Oysters are another ecosystem engineer, having major impacts on coastal ecosystems (Ruesink et al 2005). Not only are the oysters food for fish and invertebrates, they also improve water quality by filtration and provide habitat by creating biogenic reefs. These reefs influence water flow, which alters sediment deposition, consolidation, and stabilization. Thus, oysters can have disproportionately high impacts on the ecosystem, although impacts vary by species. Two important species will be considered here:
- The Olympia oyster (Ostrea lurida); native to Puget Sound but became commercially unviable due to overharvesting in the late 1800s, and despite a century of negligible harvesting, it remains commercially unviable to this day (Trimble et al 2009).
- The non-native Pacific oyster (Crassostrea gigas); commercially replaced O. lurida in 1928 and is now Washington’s most valuable shellfish resource (Dethier 2006).
The lack of recovery by native O. lurida is partially due to competition with C. gigas and other non-native and invasive species (Trimble et al 2009). Interspecific competition reduced Olympia oyster survival with C. gigas growing at twice the rate of native O. lurida (Buhle and Ruesink 2009; Trimble et al 2009). Fouling organisms, most of them non-native, kill or reduce food access to O lurida. The removal of fouling organisms, doubles the chance that O. lurida will survive and improves its growth (Trimble et al 2009). One particular invasive non-native of note that affects both O. lurida and C. gigas is the oyster drill (Ocinebrina inornata). This species was introduced before 1965 with shipments of Pacific oysters, is now established and widespread in Willapa Bay and is a significant pest of oyster aquaculture (Buhle and Ruesink 2009). Where drills are present they reduce overall survival of both oyster species, killing on average 0.33 (SE = 0.08) Pacific oysters, and 0.16 (SE = 0.04) Olympia oysters per drill per week, dependent upon prey density.
Placeholder: Green crabs, tunicates
State: Invasion of Freshwater/Riparian Ecosystems
Currently, the 2010 Washington State Noxious Weed list identifies 26 invasive non-native freshwater plant species occurring throughout Washington. These species are classified into three major classes:
- Class A- Composed of 7 invasive non-native species (2 are considered in the 50 priority species/guilds) with limited distribution in Washington and eradication is required by law.
- Class B- Composed of 14 invasive non-native species (4 are considered in the 50 top priority species/guilds), which are presently established in limited portions of the state, with containment as the primary goal.
- Class C- Composed of 5 invasive non-native species (None are considered in the 50 top priority species/guilds) that are widespread in Washington, with flexibility of control at the local level.
The Washington Department of Fish and Wildlife maintains a watch list of aquatic nuisance species of Washington’s marine and freshwaters. These non-native species are considered to have a high risk of becoming invasive and are separated into species of primary concern—those considered to have the highest level of environmental risk, and secondary concern—considered to have a lower level of environmental risk. According to the 2008 watch list, of the freshwater animals, 3 of 14 primary and 12 of 39 secondary species of concern are currently present in Washington. All but two of the primary freshwater species of concern overlaps with the top 50 priority species/guilds listed by the Washington Invasive Species Council. However, only one of the freshwater secondary species of concern overlaps with the top 50 priority species.
Impacts
Riparian zones are significant because of their ameliorating influence on aquatic ecosystems. These zones are unique ecological hotspots; instrumental in providing shelter and food for aquatic organisms, stream temperature regulation, maintaining healthy water quality by filtering contaminants and stabilizing the soil (Gregory et al. 1991; Naiman 2005). When invasive non-native species displace natives within the riparian zone, these introduced species have the potential to cause long-term cascading changes in the structure and functioning of both the riparian zone and adjacent aquatic habitat. A study by Urgenson et al (2009) found the invasive non-native giant knotweed (Polygonum sachalinense) caused such changes in community function and structure of western Washington riparian zones. Richness and abundance of native herbs, shrubs, and juvenile trees were negatively correlated with knotweed density, with a 70% reduction of native leaf litter mass. Knotweed litter has a carbon:nitrogen ratio of 52:1, which is 38-58% higher than that of native woody species. Knotweed invasion, with its litter of lower nutritional quality could affect the productivity of macro-invertebrate communities and in turn, the fish and other animals that use these invertebrates as a primary food source. Other effects of knotweed, such as decline in regeneration of red alder (a nitrogen fixer) and conifers, have important implications for nitrogen cycling and amount of large woody debris respectively.
Washington State lake ecosystems have an invasion history involving the introduction and establishment of numerous plants and animals. One species of special importance within the Puget Sound Basin is crayfish. Crayfish are a keystone species capable of effecting changes in primary productivity, food web dynamics, water quality, and biodiversity (Mueller 2007). Washington State has one native species, the signal crayfish (Pacifastacus leniusculus). However, two invasive species in Washington have been documented; in the year 2000, an invasive species, the red swamp crayfish (Procambarus clarkia) was discovered (Mueller 2002) and 2007 marked the first sighting of the northern crayfish (Orconectes virilis) (Larson and Olden 2008). As of 2008, of 58 lakes surveyed in the Puget Sound region, P. clarkia was found in ten and O. virilis was found in three (Larson and Olden 2008). The lakes that are invaded by P. clarkia, are clustered near schools which use crayfish in their science programs whereas the lakes invaded by O. virilis were all near golf courses, with ponds at golf courses oftentimes being stocked with crayfish for aquatic macrophyte control (Larson and Olden 2008). The close association between the schools and golf courses with the invaded lakes provides strong support for implicating them as introduction pathways for crayfish.
Another introduced species within the lake system is the Chinese mystery snail (Bellamya chinensis). These snails were introduced 40 years ago and are now broadly distributed in hundreds of lakes that historically supported relatively few native snails (Olden et al 2009). The Chinese mystery snail has now become a food source for both the native and invasive species of crayfish. Interestingly, the native crayfish is able to consistently handle and consume the snails at a faster pace, outcompeting both species of invasive crayfish for this novel food source (Olden et al 2009). Even so, the invasive red swamp crayfish still outnumbers the native signal crayfish by more than 2 to 1 where both species co-occur (Mueller 2007). Thus, the likelihood that red swamp crayfish will alter freshwater aquatic ecosystems in the Pacific Northwest is high (Moore 2006).
Freshwater fish species are introduced around the world due to demands for aquaculture (39%), ornamental fish (21%), modification of wild stocks (17%), sport fishing (12%), accidentally (8%) and biocontrol/engineering (6%) (Gozlan et al 2010). In western North America in particular, a variety of non-native fishes and the bullfrog (Rana catesbeiana) have been widely introduced, mainly for aquaculture and sport fishing (Adams 1999). One major impact from these introductions is the loss or decline of native amphibian species. Amphibian species richness is significantly lower at ponds having non-native predatory fish present than at either non-predatory or fish-free ponds (Hecnar and M’Closkey 2001). Even in otherwise relatively pristine high mountain lakes, lower amphibian diversity occurs in lakes harboring introduced trout, with long-toed salamanders and pacific treefrogs most negatively impacted (Bull 2002).
Non-native fish species facilitate the viability of another non-native freshwater predator, the bullfrog (citation). The bullfrog is a formidable predator, with large specimens capable of preying upon small birds, young snakes, crayfish, other frogs, and minnows. Non-native fish prey upon native macroinvertebrates thus indirectly facilitating the survival of bullfrog tadpoles (Adams et al 2003). Further, in pond surveys the best predictors of bullfrog abundance were the presence of non-native fish. However, when comparing the effects of non-native fishes and bullfrogs on red-legged frogs in the lowlands of Western Washington, red-legged frogs were significantly impacted more by non-native fishes than bullfrogs (Adams 1999). Thus, in the Pacific Northwest, non-native fish may pose a greater conservation concern than bullfrogs, at least for amphibians (Richter and Azous 1995; Adams 1999; Adams et al 2003).
Place holder: Millfoil is another important invasive in freshwater system. Himalayan blackberry, Scotch broom
1. Summary, Uncertainties and Information Gaps
Approximately 700 invasive species occur near or in the Puget Sound/Georgia Basin, many of which have become established in our native ecosystems (Washington Invasive Species Council 2009). With so many species involved, it becomes necessary to prioritize control efforts based upon ecological and economic impact. However, prioritization is no easy task considering multiple taxa and habitat types are involved and interaction/facilitation between species occurs. Some comparative studies have been attempted, but are far from being comprehensive (Adams 1999). Ranking systems appear to be useful for more comprehensive prioritization (Randall et al. 2008), although they are based on expert opinion and current knowledge. This is the method used by NatureServe (2004) and multiple states, including Washington. The assessment tool used by the Washington Invasive Species Council allows invasive non-native species to be ranked according to their ecological impact and the likelihood of Washington state agencies being able to effectively implement prevention measures or conduct early action on a species.
References
Adams, M. J. 1999. Correlated Factors in Amphibian Decline: Exotic Species and Habitat Change in Western Washington. The Journal of Wildlife Management 63:1162-1171.
Adams, M. J., C. A. Pearl, and R. Bruce Bury. 2003. Indirect facilitation of an anuran invasion by non-native fishes. Ecology Letters 6:343-351.
Boyce, R. 2009. Invasive Shrubs and Forest Tree Regeneration. Journal of Sustainable Forestry 28:1-2.
Britton-Simmons, K. H., S. Wyllie-Echeverria, E. K. Day, K. P. Booth, K. Cartwright, S. Flores, C. C. Garcia, T. L. Higgins, C. Montanez, and A. Rames. 2010. Distribution and Performance of the Nonnative Seagrass Zostera japonica across a Tidal Height Gradient on Shaw Island, Washington. PACIFIC SCIENCE 64:187-198.
Buchanan, J. B. 2003. Spartina invasion of Pacific coast estuaries in the United States: implications for shorebird conservation. BULLETIN- WADER STUDY GROUP 100:47-49.
Buhle, E. R. and R. J.L. 2009. Impacts of invasive oyster drills on olympia oyster (Ostrea lurida carpenter 1864) recovery in Willapa Bay, Washington, United States. J. Shellfish Res. Journal of Shellfish Research 28:87-96.
Bull, E. L. and D. B. Marx. 2002. Influence of Fish and Habitat on Amphibian Communities in High Elevation Lakes in Northeastern Oregon. Northwest science : official publication of the Northwest Scientific Association. 76:240-248.
Carlton, J. T. 2000. Global Change and Biological Invasions in the Oceans.in H. A. Mooney and R. J. Hobbs, editors. Invasive species in a changing world. Island Press, Washington, D.C.
Carlton, J. T. and G. M. Ruiz. 2005. Vector science and integrated vector management in bioinvasion ecology: conceptual frameworks.in H. A. Mooney and I. C. f. S. S. C. o. P. o. t. Environment., editors. Invasive alien species : a new synthesis. Island Press, Washington, DC.
Colunga-Garcia, M., R. A. Haack, and A. O. Adelaja. 2009. FOREST ENTOMOLOGY - Freight Transportation and the Potential for Invasions of Exotic Insects in Urban and Periurban Forests of the United States. Journal of economic entomology. 102:237.
Dale, V. H. and W. M. Adams. 2003. Plant reestablishment 15 years after the debris avalanche at Mount St. Helens, Washington. The Science of The Total Environment 313:101-113.
Dethier, M. N. 2006. Native Shellfish in Nearshore Ecosystems of Puget Sound. Report No. 2006-04, Puget Sound Nearshore Partnership, Seattle, WA.
Dethier, M. N. and S. D. Hacker. 2005. Physical Factors vs. Biotic Resistance in Controlling the Invasion of an Estuarine Marsh Grass. Ecological Applications 15:1273-1283.
Ellison, A. M., M. S. Bank, B. D. Clinton, E. A. Colburn, K. Elliott, C. R. Ford, D. R. Foster, B. D. Kloeppel, J. D. Knoepp, G. M. Lovett, J. Mohan, D. A. Orwig, N. L. Rodenhouse, W. V. Sobczak, K. A. Stinson, J. K. Stone, C. M. Swan, J. Thompson, B. Von Holle, and J. R. Webster. 2005. Loss of Foundation Species: Consequences for the Structure and Dynamics of Forested Ecosystems. Frontiers in Ecology and the Environment 3:479-486.
Gozlan, R. E., J. R. Britton, I. Cowx, and G. H. Copp. 2010. Current knowledge on non-native freshwater fish introductions. Journal of Fish Biology 76:751-786.
Gregory, S. V., F. J. Swanson, W. A. McKee, and K. W. Cummins. 1991. An ecosystem perspective of riparian zones. Bioscience 41:540-551.
Grevstad, F. S., D. R. Strong, D. Garcia-Rossi, R. W. Switzer, and M. S. Wecker. 2003. Biological control of Spartina alterniflora in Willapa Bay, Washington using the planthopper Prokelisia marginata: agent specificity and early results. Biological Control 27:32-42.
Grosholz, E. 2002. Ecological and evolutionary consequences of coastal invasions. Trends in Ecology & Evolution 17:22-27.
Gurevitch, J. and D. K. Padilla. 2004. Are invasive species a major cause of extinctions? Trends in Ecology & Evolution 19:470-474.
Hacker, S. D. and M. N. Dethier. 2006. Community modification by a grass invader has differing impacts for marine habitats. Oikos 113:279-286.
Hacker, S. D., D. Heimer, C. E. Hellquist, T. G. Reeder, B. Reeves, T. J. Riordan, and M. N. Dethier. 2001. A Marine Plant (Spartina Anglica) Invades Widely Varying Habitats: Potential Mechanisms of Invasion and Control. Biological Invasions 3:211-217.
Harrington, T. B. and S. H. Reichard. 2007. Meeting the challenge: invasive plants in Pacific Northwest ecosystems.
Hecnar, S. J. and R. T. M'Closkey. 1997. The effects of predatory fish on amphibian species richness and distribution. Biological conservation. 79:123.
Hobbs, R. J. 2000. Land-Use Changes and Invasions.in H. A. Mooney and R. J. Hobbs, editors. Invasive species in a changing world. Island Press, Washington, D.C.
Hulme, P. E. 2009. Trade, transport and trouble: managing invasive species pathways in an era of globalization. Journal of Applied Ecology 46:10-18.
Jenkins, P. T. and H. A. Mooney. 2006. The United States, China, and invasive species: present status and future prospects.
Larson, E. R. and J. D. Olden. 2008. Do schools and golf courses represent emerging pathways for crayfish invasions? Aquatic Invasions 3:465-468.
Lawrence, D. J. and J. R. Cordell. 2010. Relative contributions of domestic and foreign sourced ballast water to propagule pressure in Puget Sound, Washington, USA. Biological Conservation In Press, Corrected Proof.
Moore, J. W. 2006. Freshwater invertebrates-red swamp crayfish Procambarus clarkii, Rusty crayfish Orconectes rusticus, Virile crayfish Orconectes virilis.in P. D. Boersma, S. H. Reichard, and A. N. Van Buren, editors. Invasive species in the Pacific Northwest. University of Washington Press, Seattle, WA.
Mueller, K. W. 2002. First record of the red swamp crayfish, Procambarus clarkii (Girard, 1852) (Decapoda, Cambaridae), from Washington State, USA. Crustaceana 75:94
Mueller, K. W. 2007. NOTES - STATUS OF THE CRAYFISH STOCKS IN PINE LAKE, KING COUNTY, WASHINGTON FIVE YEARS AFTER THE DISCOVERY OF THE INVASIVE RED SWAMP CRAYFISH PROCAMBARUS CLARKII (GIRARD, 1852). Journal of freshwater ecology. 22:351.
Murcia, C. 1995. Edge effects in fragmented forests: implications for conservation. Trends in ecology & evolution. 10:58.
NatureServe. 2004. NatureServe explorer an online encyclopedia of life. Association for Biodiversity Information and Natural Heritage Network, Arlington, Va.
Naiman, R.J., Décamps, H., and McClain, M.E. 2005. Riparia: ecology, conservation, and management of streamside communities. Elsevier Academic Press, San Diego, California.
Olden, J. D., E. R. Larson, and M. C. Mims. 2009. Home-field advantage: native signal crayfish (Pacifastacus leniusculus) out consume newly introduced crayfishes for invasive Chinese mystery snail (Bellamya chinensis). Aquatic Ecology 43:1073-1084.
Parks, C. G., S. R. Radosevich, B. A. Endress, B. J. Naylor, D. Anzinger, L. J. Rew, B. D. Maxwell, and K. A. Dwire. 2005. Natural and land-use history of the Northwest mountain ecoregions (USA) in relation to patterns of plant invasions. Perspectives in Plant Ecology, Evolution and Systematics 7:137-158.
Phillips, C. H., K. D. Anderson, and T. O. Ketel. 2008. Spartina Eradication Program 2007 Progress Report. Washington State Department of Agriculture.
Piel, F., M. Gilbert, C. De Cannière, and J.-C. Grégoire. 2008. BIODIVERSITY RESEARCH: Coniferous round wood imports from Russia and Baltic countries to Belgium. A pathway analysis for assessing risks of exotic pest insect introductions. Diversity & Distributions 14:318-328.
Pimentel, D., L. Lach, R. Zuniga, and D. Morrison. 2000. Environmental and Economic Costs of Nonindigenous Species in the United States. BIOSCIENCE -WASHINGTON- 50:53-65.
Ramsay, M., N. Salisbury, and S. Surbey. 2004. A Citywide Survey of Habitats on Public Land in Seattle, a tool for urban restoration planning and ecological monitoring.in 16th International Conference, Society for Ecological Restoration, Victoria, Canada.
Randall, J. M., L. E. Morse, N. Benton, R. Hiebert, S. Lu, and T. Killeffer. 2008. The Invasive Species Assessment Protocol: A Tool for Creating Regional and National Lists of Invasive Nonnative Plants That Negatively Impact Biodiversity. Invasive Plant Science and Management 1:36-49.
Reichard, S. H. and P. White. 2001. Horticulture as a Pathway of Invasive Plant Introductions in the United States. Bioscience 51:103-113.
Richter, K. O. and A. L. Azous. 1995. Amphibian occurrence and wetland characteristics in the Puget Sound Basin. Wetlands : the journal of the Society of the Wetlands Scientists.:305.
Ruesink, J. L., H. S. Lenihan, A. C. Trimble, K. W. Heiman, F. Micheli, J. E. Byers, and M. C. Kay. 2005. Introduction of non-native oysters: Ecosystem effects and restoration implications. Annual Review of Ecology Evolution and Systematics 36:643-689.
Ruiz, G. M. and J. T. Carlton. 2003. Invasive species : vectors and management strategies. Island Press, Washington, DC.
Seattle Urban Nature Project. 2006. Citywide Habitat Assessment: Interim Report. Seattle, WA.
Semmens, B. 2008. Acoustically derived fine-scale behaviors of juvenile Chinook salmon (Oncorhynchus tshawytscha) associated with intertidal benthic habitats in an estuary. Canadian Journal of Fisheries and Aquatic Sciences 65:2053-2062.
Simkanin, C., I. Davidson, M. Falkner, M. Sytsma, and G. Ruiz. 2009. Intra-coastal ballast water flux and the potential for secondary spread of non-native species on the US West Coast. Marine Pollution Bulletin 58:366-374.
Trimble, A. C., J. L. Ruesink, and B. R. Dumbauld. 2009. Factors preventing the recovery of a historically overexploited shellfish species, Ostrea lurida Carpenter 1864. JOURNAL OF SHELLFISH RESEARCH 28:97-106.
Urgenson, L. S., S. H. Reichard, and C. B. Halpern. 2009. Community and ecosystem consequences of giant knotweed (Polygonum sachalinense) invasion into riparian forests of western Washington, USA. Biological Conservation 142:1536-1541.
van Kleunen, M., W. E, and F. M. 2010. A meta-analysis of trait differences between invasive and non-invasive plant species. Ecology Letters 13:235-245.
Washington Invasive Species Council. 2009. Annual Report. Seattle.
Westphal, M., M. Browne, K. MacKinnon, and I. Noble. 2008. The link between international trade and the global distribution of invasive alien species. Biological Invasions 10:391-398.
Williams, S. L. 2007. Introduced species in seagrass ecosystems: Status and concerns. Journal of Experimental Marine Biology and Ecology 350:89-110.
With, K. A. 2002. The Landscape Ecology of Invasive Spread. Conservation Biology 16:1192-1203.

