Jen McIntyre, a toxicologist at Washington State University’s Puyallup Research and Extension Center, has been studying the effects of polluted stormwater on fish for nearly 20 years. Investigating the effects of 6PPD-Q, she and her fellow researchers turned their attention to the vascular system, which carries oxygen and nutrients throughout the body. After all, the outward symptoms of gasping for air point to a lack of oxygen that could trigger neurological effects, including loss of physical control.
One study from McIntyre’s lab revealed that the 6PPD-Q somehow triggers leakage from blood vessels into the tissues — including the brains of affected fish. Normally, the brain is protected from invasion by tightly linked cells, creating a blood-brain barrier. But leakage of foreign substances into the brain — as revealed by infusing a special dye into the fish — could produce serious repercussions, according to a report by Stephanie Blair, published in the Canadian Journal of Fisheries and Aquatic Sciences. The researchers also noted leakage in the gills, which could affect respiration.
“We think the brain is important because that is the source of everything the body does,” McIntyre said. “If chemical soups can be released into the brain, that could cause all sorts of problems.”
Picking up on the idea of vascular leakage, Justin Greer, a toxicologist with the U.S. Geological Survey in Seattle, began studying cellular responses to 6PPD-Q in the tissues of newly hatched coho salmon by focusing on genes activated and deactivated by chemical exposure. Messenger RNA, a key to the production of proteins and enzymes, was fully transcribed in the exposed fish, allowing a comparison to unexposed fish. Some 1,822 genes found to be involved in the chemical response were sorted into seven groups, each having identified effects on the fish.
For example, one powerful effect revealed by the study involved the blood-brain barrier, a critical component of a healthy nervous system. Greer found that 6PPD-Q disrupted the production of proteins regulating the tightness between endothelial cells that line blood vessels and the blood-brain barrier. The effect was to loosen the spaces between the cells and allow foreign chemicals to invade the brain, as well as other tissues.
The study, published in the journal Environmental Science and Technology July 19, 2023y, also found alterations in the production of enzymes influencing inflammation and related cellular functions that also can affect the blood-brain barrier. In addition, proteins involved in the maintenance of the blood-brain barrier were affected, along with blood-related pathways such as coagulation. All these findings lead to an increased potential for leakage into the brain, organs and other tissues.
Effects on the brain may offer a general explanation for the symptoms, McIntyre noted, but researchers need to know more about how the brain is affected, as well as the steps leading from initial absorption of the chemical. It is possible that the leakage from blood vessels is a “downstream effect” of the toxic response during an enzymatic interplay within the body. Studies are ongoing to see if 6PPD-Q is acting directly on the cells or, more likely, setting off biochemical reactions that can lead to adverse effects within the brain.
Energy for cellular function
Meanwhile, other researchers are focusing on biochemical pathways that produce the raw energy needed to fuel bodily functions. Normally, cells produce energy through a major metabolic pathway involving glucose from food and oxygen from respiration. But toxic substances have been known to disrupt this key metabolic pathway and prevent the cells from utilizing available oxygen, thus leading to a type of internal suffocation.
Markus Brinkmann, a toxicologist at the University of Saskatchewan in Canada, noted from experiments in his lab as well as from other reported studies that the chemical seemed to increase glucose levels, red-blood-cell counts and oxygen consumption, in some if not all fish.
“All of that to us suggested that the mechanism might be related to energy metabolism in the widest sense,” Brinkmann said.
In 2020, when news broke that UW scientists had isolated 6PPD-Q from compounds found in tires, Brinkmann was already involved in studies looking at constituents of stormwater from the city of Saskatoon. He knew that tire chemicals were a concern, and he quickly reached out to Ed Kolodziej, an environmental engineer who was leading the UW research effort.
“One thing led to the other,” Brinkmann said, adding that his interest was piqued by the “puzzling species differences in sensitivity” along with questions about how the chemical might be affecting inland salmonids, including trout species. The University of Saskatchewan’s Toxicology Center is well-suited to study these questions, he added, “and it was clear to us that we had to get involved in this research.”
In looking at cellular energetics, graduate student Hannah Mahoney conducted experiments on two laboratory-grown cell lines from rainbow trout, namely gill cells and liver cells. In gill cells, the oxygen consumption rate increased according to 6PPD-Q concentrations, presumably because the cells were no longer able to use the available oxygen efficiently. In contrast, oxygen consumption did not change in the liver cells (Environmental Science and Technology Letters).
Meanwhile, the researchers also looked at the chemical breakdown of 6PPD-Q in the body, a process called biotransformation, which produces other chemicals, known as metabolites. A measurement of 6PPD-Q and its metabolites showed that the gill cells had converted almost none of the parent compound into metabolites, but considerable biotransformation had taken place in the liver.
“We speculated that the absence of biotransformation in the gill might actually be the reason that you see those effects on the oxygen-consumption rate in the gill cell line, not the liver cell line,” Brinkmann said, adding that those findings inspired a deeper look at the effect of the chemical on the circulatory and respiratory systems.
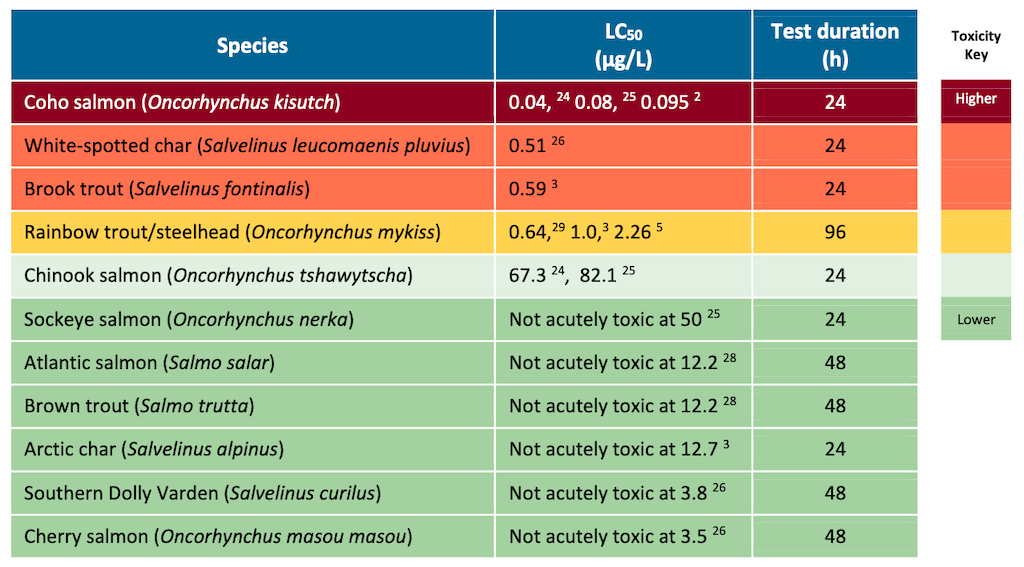
Summer Selinger, another graduate student on Brinkmann’s team, measured oxygen consumption, heart function and blood composition in two fish. One is Arctic char, shown to be insensitive to 6PPD-Q, along with rainbow trout, a highly sensitive species.
In the tolerant Arctic char, no major changes were seen in normal circulatory functions. But in the sensitive rainbow trout, oxygenated hemoglobin decreased, making oxygen less available for normal functions. At the same time, Selinger saw an increase in deoxy-hemoglobin, the form essentially searching for oxygen to carry along in the blood. And she saw an even more dramatic increase in met-hemoglobin, an undesirable form that has been chemically blocked from taking up oxygen.
These findings, yet to be published, support what observers have been seeing in the behavior of fish affected by the chemical, Brinkmann said, “almost as if they were suffocating from within.”
As another indication of physiological imbalance, the chemical also seemed to increase the heart rate in rainbow trout but not in Arctic char.
Brinkmann’s lab also conducted comparative genomic studies looking at a host of genes that became deactivated in the presence of 6PPD-Q. Researcher Catherine Roberts observed very distinct differences between sensitive and tolerant species. Using advanced modeling techniques and statistical analysis, the research team was able to organize the deactivated genes into affected biochemical pathways. The primary affected pathways seemed to be systems that regulate immune system stress and inflammation, systems that promote cell development, and systems involving cell death.
Interestingly, the modeling approach — which provides theoretical benchmark doses for the various effects — produced results that were fairly close to the effective doses shown in actual fish measured in previous experiments, said Brinkmann.
The rapid response to the chemical by living fish, along with the new genetic evidence, supports the idea that disruption could be taking place along the major biochemical pathways that produce energy for cellular function, Brinkmann said, but he stressed that neurological effects being studied in other labs could trigger a similar response.
Fitting all these pieces together — including effects on the blood-brain barrier — will require more research, Brinkmann said. New discoveries about the developmental effects on sensitive fish soon after they hatch have been setting off loud alarm bells. Genetic findings reflect some of the earliest studies on coho salmon embryos, which revealed that stormwater can reduce the length of coho and the size of their eyes. These effects could impair their ability to avoid predators.
Even low concentrations of 6PPD-Q might lead to early deformities and reduced long-term fitness in fish that somehow survive one or more exposures to the chemical. Further studies are expected to characterize these sublethal effects on a variety of fish and eventually other species, including humans.
The role of the liver in detoxification
Stark differences in sensitivity to 6PPD-Q among various species might be explained by significant differences in tissues and enzymes from one species to the next. Such differences, if they exist, could lead to varying levels of direct damage from the chemical. But another issue could be a critical factor: How quickly can an animal detoxify 6PPD-Q after it enters its body? Several studies suggest that species more tolerant to the chemical could be the ones most successful at detoxification — a primary role of the liver — thus leaving the fish less vulnerable to toxic attacks on other organs.
David Montgomery, a graduate student researcher in toxicology at the University of Saskatchewan, has been studying what happens to 6PPD-Q in the livers of various salmonids, some sensitive and some tolerant to the chemical. The more tolerant species were shown to have higher levels of metabolites, produced when chemicals in the body react with 6PPD-Q. These metabolites, created as part of the liver’s detoxification process, are less-toxic “transformational compounds.” Generally, Montgomery found the highest levels of metabolites in the most tolerant species, with metabolite levels decreasing as he tested fish with increasing sensitivity to 6PPD-Q.
The most plausible explanation for these different levels of metabolites, according to Montgomery, is that tolerant species contain more of the enzymes needed to transform 6PPD-Q to less-toxic metabolites. This is still a hypothesis, he stressed, and more investigation is needed to identify the enzymes responsible for the biotransformation.
Of mice and humans
To better understand how 6PPD-Q might affect humans and other mammals, some researchers have begun exposing mice to the chemical and looking for effects. Jing Zhang, a toxicologist at Hong Kong Baptist University, found that 6PPD-Q fed to mice was rapidly absorbed into the blood and then circulated to various parts of the body, which showed differing concentrations. The highest levels were found in the fatty adipose tissues. Peak levels in the blood were reached in less than an hour, with the chemical penetrating the blood-brain barrier within half an hour, according to the study published in the journal Science of the Total Environment.
In a limited study, patients with Parkinson’s turned out to have significantly higher concentrations of the chemical 6PPD-Q around their brains than those without the disease.
To achieve “human-relevant exposure levels,” the researchers measured the blood levels of 6PPD-Q from 30 healthy human volunteers. The mice were given oral doses of the chemical to achieve the same blood serum levels in humans. After treatment, the mice were humanely euthanized at selected intervals, and their tissues were processed to measure levels of the toxicant, according to the researchers.
The chemical was found in tissues of the kidney, lung, testis, liver, spleen, heart, and muscle. Not only did 6PPD-Q reach the highest levels in the fatty tissues, it also persisted there longer. That is consistent with the general understanding for many species that fatty tissues can store certain toxic chemicals.
Finding 6PPD-Q in lung tissue suggests that the chemical can produce effects on the lungs through contaminated food or water, even if the animal does not breathe polluted air.
The study also identified the primary metabolites of detoxification in the body, which seemed to line up with fish studies, and it concluded that fecal excretion was the primary route for expelling 6PPD-Q and its metabolites from the body, at least in mice.
In a separate study published in the journal Environmental Science and Technology, Haoqi Nina Zhao at the University of California, San Diego, found that 6PPD-Q can cross the blood-placenta barrier in pregnant mice and enter the tissues — including the brains — of developing embryos. The study also found, through a genetic assay technique, that 6PPD-Q may inhibit genes that play important roles in embryonic development, thus raising the risk of developmental abnormalities.
How closely the effects of 6PPD-Q on mice reflect the effects on humans is yet to be determined, but noninvasive studies are underway to determine the extent of human exposure to the chemical.
A South China research group found an apparent accumulation of 6PPD-Q in the cerebrospinal fluid surrounding the brains of 24 human subjects. They included patients diagnosed with Parkinson’s disease, a degenerative brain disorder that causes patients to lose control over their movements. In this limited study, patients with Parkinson’s turned out to have significantly higher concentrations of the chemical around their brains than those without the disease.
The study also removed brain cells from mice to look at the direct effects of 6PPD-Q on the neurons. Within those cells, the researchers found fibrous structures that they said were similar to structures observed in humans with Parkinson’s.
Additional work on mice revealed a disruption in the metabolic pathways within the brain cells that produced a shift in metabolites, reduced glucose uptake and decreased energy production at the cellular level. These changes are characteristic of degenerative brain disorders, the researchers said, but they acknowledged that a question remains about whether these changes are a cause or a consequence of the disorders.
“Furthermore,” the researchers noted, “there are currently no literature reports on the safe limited concentration of 6PPD-Q for humans. Addressing this gap is essential to facilitate its inclusion in safety assessments. Nevertheless, our research provides insights into the essential roles of 6PPD-Q in neurotoxicity and emphasizes that the presence of 6PPD-Q in the human brain may be a potential risk factor for Parkinson’s disease.”