Background
Determination of the significance of contamination of the Puget Sound ecosystem by toxic chemicals requires measuring the health of organisms, understanding how toxics move through the ecosystem, and estimating the risks posed by exposure to toxic chemicals. In this report we integrate some of the physical characteristics of toxics in the system with the negative effects they could cause on biota. The “threat” of toxics is dealt with separately in Section 3. Here we provide a comprehensive overview of toxics in the system, regardless of their value as an indicator of water quality. Thus, some information presented in this section comes from metrics that may not be the best indicators of water quality, but instead addresses issues of human or ecosystem health (e.g., salmon).
Toxic contaminants have been released into the Puget Sound and its watersheds for decades by human activities. Concern over the possible harmful effects of these pollutants in the ecosystem led to the creation of Washington’s Pollution Control Commission in 1945, almost 30 years before the federal Clean Water Act. The Puget Sound Water Quality Authority was established in 1985 to address pressing water quality issues, and by 1989 monitoring and assessment of water quality in Puget Sound had begun with the Puget Sound Ambient Monitoring Program (PSAMP).
The goals of PSAMP included characterizing status and trends of the condition of Puget Sound. Now called the Puget Sound Assessment and Monitoring Program, it currently exists as a consortium of regional scientists from a number of agencies who monitor and assess ecosystem health. Other ongoing toxics monitoring efforts in Puget Sound include MusselWatch (Kimbrough et al. 2008), a national program that has been active in Puget Sound since the 1980s, and King County’s Marine Monitoring Program .
The Washington Department of Ecology has evaluated and identified 17 chemicals of concern for Puget Sound (Table 1)(Hart Crowser 2007), based on threat or known harm to biota. Of these, only five chemicals have been banned nation-wide under the Toxic Substances Control Act (TSCA) since 1976. Washington State recently became the first state to ban a class of relatively new chemicals, polybrominated flame retardants (PBDEs), because of human and environmental health concerns.
Table 1. Washington Department of Ecology’s list of Chemicals of Concern. Table reprinted from Hart Crowser 2007
Chemical of Concern | Category Addressed | Harm or threat |
Arsenic | Arsenic | Associated with sediment toxicity and benthic community impairment |
Cadmium | Cadmium | Accumulation in shellfish |
Copper | Copper | Associated with sediment toxicity and benthic community impairment; affects salmonids and stream health |
Lead | Lead | Associated with sediment toxicity and benthic community impairment |
Mercury | Mercury | Target of fish consumption advice; associated with sediment toxicity and benthic community impairment |
Total PCBs (a) | PCBs | Liver lesions and reproductive impairment in fish from urban bays; associated with sediment toxicity and benthic community impairment |
Low molecular weight PAHs (b) | PAHs | Liver lesions and reproductive impairment in fish from urban bays; associated with sediment toxicity and benthic community impairment |
Carcinogenic PAHs (c) | PAHs | Liver lesions and reproductive impairment in fish from urban bays; associated with sediment toxicity and benthic community impairment |
Other high molecular weight PAHs (d) | PAHs | Accumulation in fish, birds, and mammals; associated with sediment toxicity and benthic community impairment |
Sum of DDT and metabolites € | Pesticides | Category thought to affect salmonids and stream health |
Triclopyr (f) | Pesticides | Accumulation in birds and mammals; furans associated with sediment toxicity and benthic community impairment |
Total dioxin TEQs from dioxins and furans (g) | Dioxins and furans | Category shown to accumulate in fish, invertebrates, and sediment of urban waterways at levels triggering sediment clean up activities |
bis(2-Ethylhexyl)phthalate | Phthalate esters | Accumulation in sediments, fish, and harbor seals |
Total PBDEs (h) | PBDEs | Category thought to cause reproductive impairment observed in fish from urban bays |
Nonylphenol | Hormone disrupting chemicals | Kills and reduces fitness of marine organisms |
Oil or petroleum product (i) |
| Increasing concentrations may threaten aquatic resources |
- Sum of polychlorinated biphenyl congeners
- Polyaromatic hydrocarbons: acenaphthene, acenaphthylene, anthracene, fluorene, naphthalene, and phenanthrene (per WAC 173-204-320)
- Polyaromatic hydrocarbons: benz(a)anthracene, benzo(a)pyrene, benzo(b)fluoranthene, benzo(k)fluoranthene, chrysene, dibenz(a,h)anthracene, and indeno(1,2,3-c,d)pyrene (per USEPA)
- Polyaromatic hydrocarbons: benzo(g,h,i)perylene, fluoranthene, and pyrene (WAC 173-204-320 high molecular weight PAHs not on U.S. EPA list of carcinogenic PAHs).
- DDT = Dichlorodiphenyltrichloroethane.
- Input from the project team did not reflect consensus to include this compound as currently used pesticide. Other candidates suggested by project team members included diazinon and dichlorbenil.
- TEQ = Toxicity equivalent.
- PBDEs = Polybrominated diphenyl ethers. Sum of congeners have been normalized.
- Specified as crude oil, specific refined product (e.g., diesel, gasoline, heavy fuel oil), or analytical result as TPH-D or TRPH.
Toxic contaminants are considered a priority threat in Puget Sound because they may harm the health of biota. In many cases harm can be difficult to observe; effects can be non-lethal (behavioral) or affect reproductive potential. The status of toxic contaminants in ecosystems typically is reported using a) metrics of exposure, such as the concentration of contaminant residues in tissues; b) health effects such as cancer or reproductive impairment that are known to be caused by such exposure (i.e., are “toxicopathic”); c) concentration of toxics in abiotic media such as sediments or water; d) toxicity of abiotic media; e) benthic infaunal community metrics, or f) an index value calculated from some combination of a-e. The process of “bioconcentration” of toxics from abiotic media to biota is well documented in some cases, suggesting that toxic contaminants in abiotic media can serve as a proxy for or predictor of exposure (Meador 2006).
Measuring toxic contaminants in the environment is expensive and sometimes logistically difficult, so monitoring and assessment studies or programs are challenged with targeting contaminants that pose the greatest threat based on a number of criteria including:
- level of toxicity to organisms
- types of harm caused
- persistence in the environment
- rates of bioaccumulation and biomagnification
- frequency of occurrence in the ecosystem
- spatial distribution in the ecosystem
- threats to specific taxa
Furthermore, the toxicity of a contaminant to an organism depends on the degree to which it is exposed to the chemical. Ideally, status is reported with respect to both the degree of exposure, and the effects (impacts) that exposure causes.
This section summarizes the status and trends of contaminant exposure and effects for key species to four major classes of toxic contaminant. Metrics reported here include: a) measurements of contaminant concentration in organisms’ bodies (“tissue residues”) or concentration of contaminant metabolites; b) toxicopathic effects (e.g., liver disease and various measures of reproductive impairment); c) concentration of toxics in sediments, primarily as a source of and proxy for biotic exposure; and d) a multimetric toxics-related index of sediment health.
The focus of this section is on toxic contaminants as they relate to biotic exposure and effects. Various species have been used over the years as indicators of toxics status and trends, based on key life history characteristics designed to evaluate the presence, fate, and transport of toxics in the complete food web. Key life history characteristics include:
- where the organism lives (its habitat, e.g., benthic vs pelagic)
- trophic level
- longevity (long lived species have a greater potential for accumulative exposure)
- migration/residency relative to contaminated habitats
- prey or food preferences
Furthermore, the focus of this report is limited to the marine ecosystem. Evaluation of loadings and sources, such as from stormwater or atmospheric deposition, is not included.
Toxic contaminants in sediments and fish tissues have been two of the most widely monitored and assessed indicators of ecosystem health in Puget Sound. Understanding the significance of the threat posed by sediment contamination requires an understanding of the relationship between sediment pollution, biotic exposure, and the movement of contaminants from sediments to biota. The majority of data useful for a broad-scale evaluation of status and trends in both sediment and biota comes from the PSAMP long-term monitoring and assessment studies. Results from these efforts have been published primarily in the periodic Puget Sound Update series and in other state agency reports. Most PSAMP data collection methods use vetted protocols (e.g., Puget Sound Estuary Program 1989a (revised), 1989b (Revised), 1990, 1996a, 1996b) which may have been modified over time following internal agency peer review or review among PSAMP principal investigators. Reviews of PSAMP were performed by a panel of external experts in 1995 (Shen 1995) and again in 2005 by PSAMP’s Management Committee (PSAMP unpublished). In cases where agency-endorsed or other adequate processes for peer review were performed, and where procedures were vetted as above, PSAMP results from the Puget Sound Update series or other Agency reports are cited or presented here. Data or findings that fail to meet these requirements are omitted.
Status and Trends
Persistent Bioaccumulative Toxics (PBTs)
Persistent bioaccumulative toxic contaminants are a class of substances comprising primarily synthetic chemicals designed and manufactured to meet a wide range of industrial, agricultural, or residential needs. Because they are persistent and bio-accumulative, they are cause for concern when released into the environment. These chemicals generally resist physical, chemical, and metabolic breakdown, so they remain unchanged in the environment for a long period of time. Their concentration increases in the body with chronic or increasing exposure or intake, and they are toxic, causing harm to biota. Because of these characteristics, PBTs have been the focus of intense research world-wide, and large PBT databases exist for risk assessors, modelers, and regulators (Weisbrod et al. 2007).
In Puget Sound marine and estuarine waters the PBTs of primary concern are summarized by Hart Crowser (2007). Those for which broad status information exists in Puget Sound include polychlorinated biphenyls (PCBs), polybrominated diphenyl ethers (PBDEs), organo-chlorinated pesticides (OCPs) such as dichlorodiphenyltrichloroethane (DDTs), and mercury. These contaminants have been measured or monitored in a wide range of species in Puget Sound from as early as the mid 1970s to present, with consistent monitoring in several species from 1989 to present. Although polychlorinated dibenzo-p-dioxins (PCDDs), and polychlorinated dibenzofurans (PCDFs) have been detected in English sole from the most heavily contaminated embayment in Puget Sound (Elliott Bay; Sloan and Gries 2008), these compounds are considered a minor threat to apex predators such as harbor seals in Puget Sound (Ross et al. 2004) that could otherwise potentially be exposed to high PBT levels via bio-magnification.
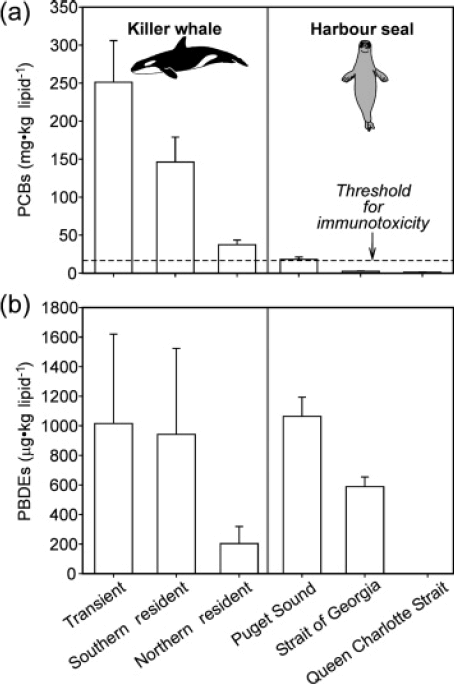
Perhaps the clearest PBT exposure-effects relationship of concern in the Puget Sound marine waters is the exposure of apex predators such as Southern Resident Killer Whales (SRKW) and harbor seals to PCBs and PBDEs (Figure 1). Hickie et al. (2007) and Ross et al. (2004) reported PCB exposure in harbor seals from Puget Sound at levels predicted to impair health, while Ross et al. (2000) described the SRKW population as the most PCB-contaminated of all cetaceans in the world. Calculations made by Hickie et al. (2007) and Ross et al. (2004) suggested that during their years of peak exposure, all members of the SRKW population were affected, and that exposure exceeded thresholds by 3 to 31 times. The authors estimated that based on PCBs alone, it would take until the year 2089 for 95% of the population to drop below the health effects threshold, given current PCB trends. Such PBT contamination is considered a risk to recovery of this population (Krahn et al. 2002).
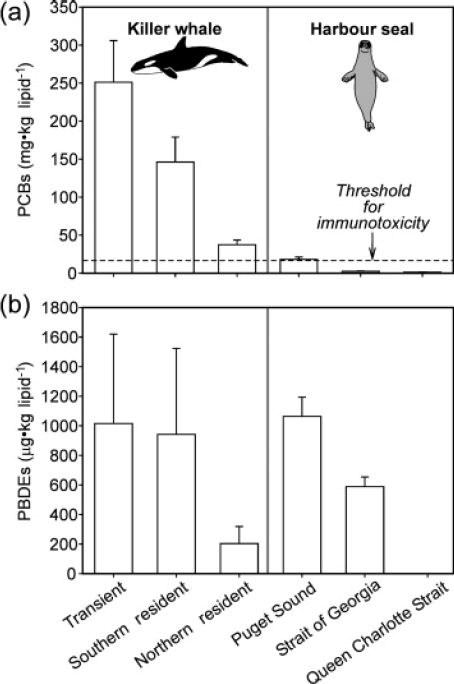
Figure 1. Persistent bioaccumulative toxics (PCBs and PBDEs) in two apex predators from the Puget Sound and Strait of Georgia, with health effects threshold for PCBs. Reprinted with permission from Ross (2006)
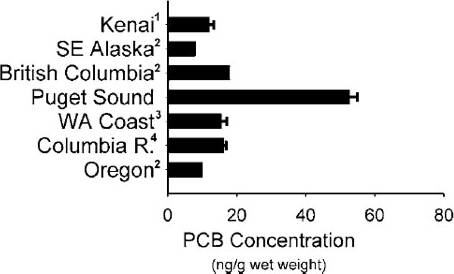
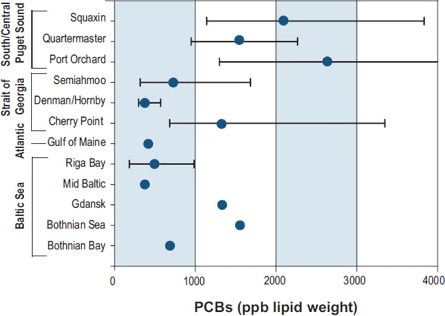
The source of PCBs to these animals is their food, primarily chinook salmon for killer whales (e.g., Krahn et al. 2007) and a mix of small pelagic and benthic fish for harbor seals (Cullon et al. 2005). O'Neill and West (2009) reported high PCB body burdens in chinook salmon that reside in Puget Sound, compared to more oceanic migrants (Figure 2) and West et al. (2008) reported high PBC burdens in Pacific herring from Central and Southern Puget Sound, compared with Southern Strait of Georgia and with herring from highly polluted regions of the Baltic Sea (Figure 3). This illustrates the importance of PBT transfer via trophic interactions and the need to understand PBT fate and transport processes in food webs.

Figure 2. Comparison of PCB tissue residues in adult Chinook salmon returning to spawn in Puget Sound and Pacific Oceanic coastal rivers. See West and O’Neill 2009 for a description of sampling location and full data citations. All samples were from adult Chinook salmon returning to natal rivers to spawn. Copyright American Fisheries Society. Used with permission.
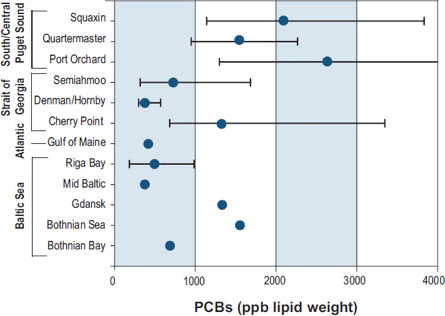
Figure 3. Comparison of PCBs among six populations of Pacific herring Puget Sound and the Georgia Basin, and Atlantic and Baltic herring. Squaxin population from South Puget Sound, Quartermaster Harbor and Port Orchard from Cental Puget Sound, and Semiahmoo, Denman/Hornby, and Cherry Point from the Southern Strait of Georgia (Reprinted from 2007 Puget Sound Update; data from West et al. 2008)
PCB exposures in chinook salmon pose a health risk to the fish, as well as to the humans that consume them. PCBs in chinook salmon (O'Neill and West 2009) exceeded an effects threshold reported by Meador (2002), indicating a threat to normal growth and maturation processes for these salmon. Furthermore, the Washington Department of Health has issued guidelines that recommend restrictions to dietary intake of these fish to protect human health .
In sediments, PBTs tend to accumulate in industrial or urbanized habitats near their sources, prompting focused attention on toxics there (Partridge et al. 2009, Puget Sound Estuary Program 1988). The Environmental Protection Agency’s Superfund program has focused sediment cleanup efforts in a number of Puget Sound’s urbanized embayments since 1980 (2007 Puget Sound Update). Overall, however, Ecology’s long-term PSAMP efforts (methods peer reviewed: Dutch et al. 2009) have reported PCB levels in sediments exceeding Washington State Sediment Quality Standards (SQS) in only 19 of over 500 stations from the full extent of Puget Sound sampled between 1997 and 2008. Washington State Sediment Quality Standards, adopted as part of Washington’s environmental regulations, define levels at which various chemicals present in sediments become harmful to marine life (WAC 173-204). All PCB exceedances were located in sediments taken from urban embayments in the Central Puget Sound basin. Data indicate that PCB concentrations in Elliott Bay sediments, where most of the exceedances have occurred, have been declining (Partridge et al. 2009). A Washington State Sediment Quality Standard does not yet exist for PBDEs, and although PBDE concentrations were lower than PCBs overall, they were concentrated in Central Puget Sound and its urbanized embayments.
Long-term Sound-wide monitoring efforts have shown that this urban PCB and PBDE sediment signal is reflected in benthic (bottom-dwelling) and demersal (near-bottom) species. Tissue residues of PCBs and PBDEs were greatest in English sole (benthic), rockfish (demersal) and lingcod (demersal) from Elliott Bay, Commencement Bay, and Sinclair Inlet, or from other Central Puget Sound urban or near-urban locations (as reported in 2007 Puget Sound Update). PCB residues in blue mussels were greatest in Central Puget Sound locations (Kimbrough et al. 2008). These studies demonstrate the relationship between benthic (or benthic-feeding) species and the contaminant-condition of their environment.
Although pelagic (open-water) species may not have direct trophic connections with the sediment-contaminated benthic food web, pelagic food web species in urbanized waters exhibited high levels of exposure to PBTs. Pacific herring (West et al. 2008), Chapter2a.Salmonids#chinookanchor|chinook salmon]] (O'Neill and West 2009), and harbor seals (Ross et al. 2004) that reside in Puget Sound conform to this pattern. PCB and PBDE tissue residues were consistently greatest in individuals of these three species from the Central or Southern Puget Sound Basins, compared with conspecifics from the Strait of Georgia, Strait of Juan de Fuca, or Pacific Ocean. As noted previously, PCB and PBDE tissue residues exceeded health effects thresholds in salmon and harbor seals.
PBDEs have only relatively recently been added to tissue monitoring and assessment programs in Puget Sound. Using archived tissue samples, West and O'Neill (2007) observed 80 ng/g Total PBDEs (wet wt) in herring from Central Puget Sound in 2001, roughly one-half the concentration of Total PCBs reported for the same samples from (West et al. 2008).
Polycyclic Aromatic Hydrocarbons (PAHs)
PAHs are derived from fossil fuels, and are typically produced via combustion of these fuels (pyrogenic) or occur as constituents of petroleum (petrogenic). Most of these chemicals exist naturally, but their presence in the environment becomes problematic when they are concentrated to toxic levels by human activities. Many PAHs are persistent in the environment, however they are typically metabolized by vertebrates when exposed to relatively low concentrations, and therefore do not tend to accumulate in their bodies. For this reason, food web magnification of PAHs for apex predators is of less concern than for PBTs.
However, both exposure and effects measures from biota indicate that PAHs represent a serious threat to the health of some Puget Sound biota. PAHs in blue mussels from seven of 14 sites in Puget Sound waters were termed “high” (at or above the 85th percentile for all 263 stations nationwide in at least half the years sampled between 1986 and 1991) by the national Mussel Watch Program (O'Connor 2002). Currently, the PAH status of mussels from eight of 10 stations in Puget Sound is rated either “medium” or “high” (Kimbrough et al. 2008), with a number of locations that met or exceeded comparable mussel samples taken in highly urbanized areas of the Baltic Sea. Tissue residues of PAHs in blue mussels could originate from capturing and consuming PAH-laden particles derived from nearby sediments (Baumard et al. 1999). This hypothesis is supported by the observation that PAHs in Puget Sound mussels are typically greatest in urban sites (Kimbrough et al. 2008).
Because PAHs are metabolized by vertebrates, measuring the exposure of fish, birds and mammals is less straightforward than measuring PBT tissue residues. Metabolites of PAH compounds can be measured in fish bile (Krahn et al. 1984), and these so-called biliary Fluorescing Aromatic Compounds (FACs) have been monitored in English sole, rockfish and herring as a semi-quantitative measure of PAH exposure in these species in Puget Sound (West et al. 2001). In the benthic or demersal fish species biliary FACs were consistently greatest in fish taken from urbanized embayments.
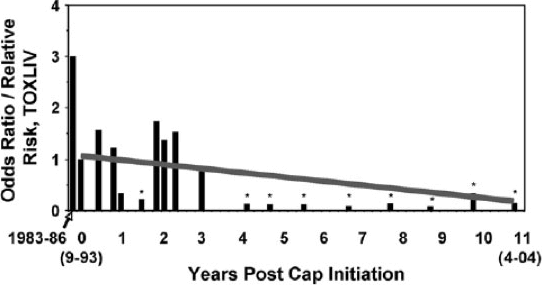
PAHs are linked to a number of toxicopathic fish diseases. English sole develop degenerative liver disease when exposed to PAHs in the sediments where they feed (Myers et al. 1990, Myers et al. 1991). Other effects include interruption in growth, and reproductive impairments (Johnson et al. 2002). Myers et al. (2008) documented the recovery of health among English sole in Eagle Harbor, a highly PAH-contaminated Superfund site, where prevalence of PAH-induced liver disease dropped from 80% to 5% over a ten year period during remediation, which included sequestration of PAHs with sediment capping (Figure 4).
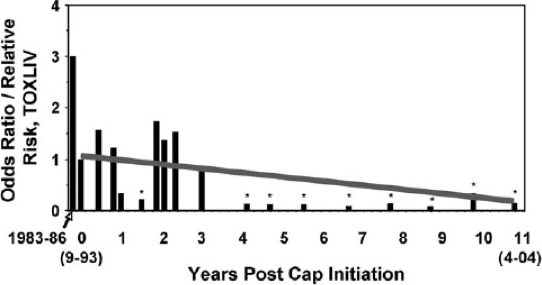
Figure 4. Temporal trend in liver disease of English sole from a sediment-remediated site in Eagle Harbor, Washington. Reprinted with permission from Myers et al. 2008.
PAH-induced liver disease has been tracked in English sole by PSAMP for 20 years in eight Puget Sound locations. This tracking study uses protocols developed to monitor histopathological health metrics in fish, including toxicopathic liver disease (Puget Sound Estuary Program 1987), which are regularly reviewed by a contract pathologist. Recent results from this tracking study, reported in the 2007 Puget Sound Science Update, include the following:
- liver disease in English sole is associated primarily with Puget Sound’s highly contaminated urban embayments near Seattle (Elliott Bay), Tacoma (Thea Foss Waterway), and Everett (Port Gardner). The risk of developing liver disease in these areas was two to six times that of normal background
- the risk of developing PAH-induced liver disease has remained low and unchanged at six of eight long-term stations, and has declined significantly in English sole from Elliott Bay (Seattle).
- high molecular weight PAHs, the group most often associated with liver disease (Myers et al. 1991), have declined in Elliott Bay sediments (Partridge et al. 2009), and in English sole bile from Elliott Bay
Pelagic fish in Puget Sound have also exhibited exposure to PAHs. Pacific herring, a small, schooling pelagic planktivorous fish, have consistently exhibited PAH exposures in Central Puget Sound similar in magnitude to benthic (English sole) and demersal (rockfish) species from most urbanized embayments for the past 10 years (2007 Puget Sound Science Update).
The source of persistent organic pollutants such as PAHs in adult fishes is widely thought to be dietary and because PAHs are metabolized, their biliary FAC measurements tend to reflect PAH loads in prey that have been consumed recently. Pacific herring is a fully pelagic species that consumes primarily zooplankton prey, with little obvious trophic connection to contaminated sediments in Puget Sound. Small schooling pelagic planktivores such as herring may accumulate PAHs that have originally been ad- or absorbed to plankton, and then magnified among planktonic invertebrates in the food web (Wolfe et al. 2001). It has been suggested that some PAHs loaded from atmospheric deposition or other sources enter the pelagic food web directly via bioaccumulation by phytoplankton, and then are magnified through the planktonic food chain to their fish predators (Larsson et al. 2000). In Puget Sound, this may explain why pelagic species far removed, both trophically and spatially, from contaminated sediments exhibit such high exposure to PAHs, and could help to inform decisions regarding how to mitigate exposure of biota to PAHs in Puget Sound.
PAH exposure may pose a significant threat to sensitive life stages of Puget Sound biota. Links between chronic, sublethal PAH levels and health impacts in fish embryo and larval stages, as well as delayed population declines from early-life PAH exposures have been well established (Carls and Meador 2009, Peterson et al. 2003). In addition, PAHs from creosote, such as found on treated pilings, can kill embryos (Vines et al. 2000). Herring embryos exhibiting chronic mortality from at least one spawning ground in Puget Sound have experienced exposure to PAHs exceeding a PAH effects threshold (as reported in 2007 Puget Sound Science Update), however a PAH cause-and-effect link to mortality has not yet been established in Puget Sound.
Contaminants of Emerging Concern (CECs)
This group of contaminants comprises a broad range of chemical classes whose adverse effects on biota is only recently becoming apparent. They range widely in solubility, persistence, toxicity, and mode-of-action, and include such classes as perfluorinated compounds (from the creation of fluoropolymers, semiconductors, and fire-fighting foam), nonylphenol (a surfactant), bisphenol-A and phthalates, both used in plastics, and pharmaceuticals and personal care products. Many of these contaminants have endocrine disrupting capacity, and so may be reported as Endocrine Disrupting Compounds (EDCs); some are specifically estrogenic and so may be reported as xenoestrogens. Although some of these contaminants have been measured in Puget Sound fishes (West et al. 2001), and some are monitored in Puget Sound sediments (Dutch et al. 2009), many CECs currently are not measured in environmental media on a broad scale (Muir and Howard 2006). Moreover, analytical techniques for measuring tissue residues or metabolites for many CECs are lacking. Two CECs, nonylphenol (NNP) and bis(2-ethylhexyl)phthalate (DEHP) are included in Washington Department of Ecology’s Chemicals of Concern list (Hart Crowser 2007).
In Puget Sound sediments, at least one phthalate-chemical, DEHP, exceeded the Washington State sediment quality standard, and appears to be increasing (Partridge et al. 2009). DEHP is associated with a wide range of toxicopathic disease including endocrine disruption (e.g., feminization of males). English sole in Puget Sound have shown evidence of exposure to xenoestrogenic compounds, even though the causative pollutants remain unknown. Johnson et al. (2008) reported the presence of vitellogenin, a precursor to egg protein normally found only in females, in male English sole from twelve of sixteen locations sampled in Puget Sound. Moreover, both females and males from one affected population in Elliott Bay exhibited altered reproductive timing, possibly related to the unknown estrogenic pollutants (Figure 5). Based on spatial patterns in the fish impairment, these authors hypothesized that the source of xenoestrogens to these fish was industrial discharges, surface runoff, or sewage, and discussed the most likely causative pollutants.
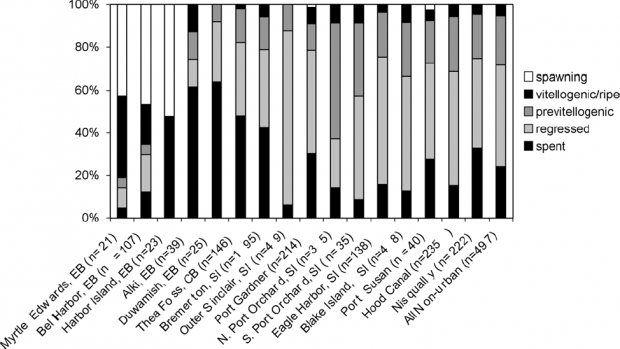
Figure 5. Unusual reproductive timing in female English sole from three Elliott Bay sites compared to 14 other Puget Sound locations. Reprinted with permission from Johnson et al. 2008.
Metals
Like PAHs, metals occur naturally in the environment. Metals become contaminants of concern when they are altered chemically or redistributed in the environment in ways that make them more available or toxic to biota. In some cases (e.g., mercury) metals may naturally occur in biota in a magnitude great enough to cause concern for humans consuming them (Barghigiani and DeRanieri 1992).
Metals have been monitored Sound-wide in sediments (Dutch et al. 2009) and fish tissue (West et al. 2001) since 1989, and in blue mussels since 1986 (Kimbrough et al. 2008). Metal accumulation in mussels has been unremarkable, except that the greatest tissue residues of mercury, nickel, and lead occurred in highly urbanized areas, suggesting anthropogenic contributions. “Medium” concentration of a number of metals was reported from locations with the greatest exposure to oceanic waters, far removed from human activities, suggesting accumulation of natural sources.
The Sediment Quality Triad
The Sediment Quality Triad Index (SQTI) is a multi-metric index developed to describe the degradation of sediment condition by toxic contaminants (Chapman 1990). Because the SQTI incorporates toxic contaminants from a broad range of classes, it is presented separately in this section. The SQTI combines the results from pollutant concentration measures, toxicity studies (exposing sensitive organisms to sediments or their extracts), and analysis of the composition of the infaunal community in sediments. This last measure is typically based on best professional judgment, and integrates nine different measures of community structure and the presence/absence of pollutant tolerant/sensitive species. A substantial advantage of the SQTI is that it examines both effects and exposure metrics, and uses a weight-of-evidence approach to integrate three important measures of sediment quality into one indicator that can be compared Puget Sound-wide.
SQTI has been used extensively in Puget Sound as an indicator of sediment health (Long et al. 2003). A seven-year comprehensive SQTI survey of 381 Puget Sound sediment stations reported that although only one percent of sediments were “degraded”, most of these sediments were in highly productive shallow-water embayments or river deltas (reported in 2007 Puget Sound Science Update). Thirty-eight percent of sediments in Puget Sound were considered of “intermediate” quality, wherein degradation was detected in one or two of the three SQTI metrics. A full, final report for this study is currently being reviewed by the Washington Department of Ecology.
Uncertainties.
One important uncertainty concerns the linkage between the status and trends of toxic contaminants in the ecosystem and the associated population-level effects on biota. Health-effects thresholds are lacking for the great majority of toxic contaminants monitored in Puget Sound, especially for complex mixtures of chemicals. Constructing models that predict population-level effects from lethal or sub-lethal effects of single contaminants or mixtures is a challenge, because such models often carry a great deal of uncertainty that can result in wide-ranging outcomes. Except for oil spills or other episodic events, observations of mortality directly attributable to toxic contaminants are rare. A singular exception to this in Puget Sound is pre-spawning mortality of coho salmon in urban streams (Collier et al. 2004), a phenomenon widely attributable to road-based contaminants. In this case Spromberg and Scholz (2009) predict extirpation of coho spawning runs over decadal time scales in urbanized streams.
In addition, recent findings on the susceptibility of eggs and larvae of Pacific herring to fossil fuel-derived PAH compounds (Carls et al. 1999, Incardona et al. 2009) combined with field studies that demonstrate exposure of their embryos to PAHs in Puget Sound (as reported in 2007 Puget Sound Update) show the risk of mortality to this sensitive life stage from exposure to chemical pollutants in Puget Sound. However, because such mortalities are extremely difficult to observe or measure in the wild, they currently are not used in decision-making.
Uncertainties unique to status and trends of monitoring data include shifting methodologies and study designs over long time periods. For example, PCBs reported from some studies in this document have been analyzed using a range of methodologies including mixtures (Aroclor) analysis and congener-based methods. Careful evaluation of all methods, including those for biological covariates, must be made when comparing these data across studies, and when applying threshold criteria.
Summary
Human activities have resulted in the introduction or elevation of toxic contaminants into Puget Sound. These include Persistent Bio-accumulative Toxics such as PCBs, PBDEs and DDTs, chemicals derived from fossil-fuels (PAHs), various metals, and Contaminants of Emerging Concern, including Endocrine Disrupting Compounds and pharmaceuticals and personal care products. In Puget Sound, a number of PBT chemicals are present in apex predators such killer as whales and harbor seals and in their primary food sources (salmon and herring) in concentrations that may harm their health and impair recovery of populations that are depressed. For most PBTs, the highest concentrations occur in sediments and biota from the Central Puget Sound and its urbanized embayments, or localized urbanized shorelines in other Puget Sound basins. PAH monitoring of mussel tissue has caused Puget Sound to be characterized as a hot spot for that class of contaminants, relative to other urban areas in the nation. PAH chemicals have also been detected in fish bile and identified as a causative factor in liver disease in English sole in Puget Sound waters. Juvenile life stages of fish may be particularly susceptible to PAH toxicity. Reproductive effects of endocrine-disrupting compounds have been detected in benthic Puget Sound fish but the consequences of exposure at the population level and long-term trends are not known.
References
Barghigiani, C., and DeRanieri, S. 1992. Mercury content in different size classes of important edible species of the Northern Tyrrhenian Sea. Marine Pollution Bulletin 24(2): 114-116.
Baumard, P., Budzinski, H., Garrigues, P., Narbonne, J.F., Burgeot, T., Michel, X., and Bellocq, J. 1999. Polycyclic aromatic hydrocarbon (PAH) burden of mussels (Mytilus sp.) in different marine environments in relation with sediment PAH contamination, and bioavailability. Marine Environmental Research 47(5): 415-439.
Carls, M.G., and Meador, J.P. 2009. A Perspective on the toxicity of petrogenic PAHs to developing fish embryos related to environmental chemistry. Human and Ecological Risk Assessment: An International Journal 15(6): 1084 - 1098.
Carls, M.G., Rice, S.D., and Hose, J.E. 1999. Sensitivity of fish embryos to weathered crude oil: Part I. Low-level exposure during incubation causes malformations, genetic damage, and mortality in larval Pacific herring (Clupea pallasi). Environmental Toxicology and Chemistry 18(3): 481-493.
Chapman, P.M. 1990. The sediment quality triad approach to determining pollution-induced degradation. Science of The Total Environment 97-98: 815-825.
Collier, T.K., Labenia, J., Myers, M.S., and Scholz, N.L. 2004. Abstracts from the twelfth international symposium on pollutant respsonses in marine organisms - mechanisms of toxicity - organics: Extreme PRIMO: premature mortality of adult coho salmon associated with stormwater discharges. Marine Environmental Research 58(2-5): 359-369.
Cullon, D.L., Jeffries, S.J., and Ross, P.S. 2005. Persistent organic pollutants in the diet of harbor seals (Phoca vitulina) inhabiting Puget Sound, Washington (USA), and the Strait of Georgia, British Columbia (Canada): a food basket approach. Environmental Toxicology and Chemistry 24(10): 2562-2572.
Dutch, M., Partridge, V., Weakland, S., Welch, K., and Long, E. 2009. Quality Assurance
Project Plan: The Puget Sound Assessment and Monitoring Program Sediment Monitoring Component. Washington State Department of Ecology. Olympia, WA
Hart Crowser. 2007. Control of Toxic Chemicals in Puget Sound. Phase 1: Initial estimate of loadings. Washington Department of Ecology. Olympia, WA
Hickie, B.E., Ross, P.E., MacDonald, R.W., and Ford, J.K.B. 2007. Killer whales (Orcinus orca) face protracted health risks associated with lifetime exposure to PCBs. Environmental Science and Technology 41(18): 6613-6619.
Incardona, J.P., Carls, M.G., Day, H.L., Sloan, C.A., Bolton, J.L., Collier, T.K., and Scholz, N.L. 2009. Cardiac Arrhythmia Is the Primary Response of Embryonic Pacific Herring (Clupea pallasi) Exposed to Crude Oil during Weathering. Environmental Science & Technology 43(1): 201-207.
Johnson, L., Collier, T.K., and Stein, J.E. 2002. An analysis in support of sediment quality thresholds for polycyclic aromatic hydrocarbons (PAHs) to protect estuarine fish. Aquatic Conserv: Mar. Freshw. Ecosyst. 12: 517-538.
Johnson, L.L., Lomax, D.P., Myers, M.S., Olson, O.P., Sol, S.Y., O'Neill, S.M., West, J., and Collier, T.K. 2008. Xenoestrogen exposure and effects in English sole (Parophrys vetulus) from Puget Sound, WA. Aquatic Toxicology 88(1): 29-38.
Kimbrough, K.L., Lauenstein, G.G., Christensen, J.D., and Apeti, D.A. 2008. An assessment of two decades of contaminant monitoring in the Nation’s Coastal Zone. National Oceanic and Atmospheric Administration/National Center for Ocean Science Technical Memorandum, Silver Spring, MD.
Krahn, M.M., Hanson, M.B., Baird, R.W., Boyer, R.H., Burrows, D.G., Emmons, C.K., Ford, J.K.B., Jones, L.L., Noren, D.P., Ross, P.S., Schorr, G.S., and Collier, T.K. 2007. Persistent organic pollutants and stable isotopes in biopsy samples (2004/2006) from Southern Resident killer whales. Marine Pollution Bulletin 54(12): 1903-1911.
Krahn, M.M., Wade, P.R., Kalinowski, S.T., Dalheim, M.E., Taylor, B.L., Hanson, M.B., Ylitalo, G.M., Angliss, R.P., Stein, J.E., and Waples, R.S. 2002. Status Review of Southern Resident Killer Whales (Orcinus orca) under the Endangered Species Act. Edited by U.S.D.o. Commerce. p. 133.
Krahn, M.M., Myers, M.S., Burrows, D.G., and Malins, D.C. 1984. Determination of metabolites of xenobiotics in the bile of fish from polluted waterways. Xenobiotica 14(8): 633-646.
Larsson, P., Andersson, A., Broman, D., Nordbäck, J., and Lundberg, E. 2000. Persistent organic pollutants (POPs) in pelagic systems. Ambio 29(4-5): 202-209.
Long, E.R., Dutch, M., Aasen, S., Welch, K., and Hameedi, M.J. 2005. Spatial extent of degraded sediment quality in Puget Sound (Washington State, U.S.A.) based upon measures of the sediment quality triad. Environmental Monitoring and Assessment 111: 173-222.
Long, E.R., Dutch, M., Aasen, S., Welch, K., and Hameedi, M.J. 2003. Chemical contamination, acute toxicity in laboratory tests, and benthic impacts in sediments of Puget Sound: a summary of results of the joint 1997-1999 Ecology/NOAA survey. Publication Number 03-03-049, or Technical Memorandum NOS NCCOS CCMA 163, Washington Department of Ecology, Lacey, WA.
Meador, J. 2006. Rationale and Procedures for Using the Tissue-Residue Approach for Toxicity Assessment and Determination of Tissue, Water, and Sediment Quality Guidelines for Aquatic Organisms. Human and Ecological Risk Assessment: An International Journal 12(6): 1018 - 1073.
Meador, J.P. 2002. Use of tissue and sediment-based threshold concentrations of polychlorinated biphenyls (PCBs) to protect juvenile salmonids listed under the US Endangered Species Act. Aquatic Conservation: Marine and Freshwater Ecosystems 12: 493-516.
Muir, D.C.G., and Howard, P.H. 2006. Are There Other Persistent Organic Pollutants? A Challenge for Environmental Chemists. Environmental Science & Technology 40(23): 7157-7166.
Myers, M.S., Anulacion, B.F., French, B.L., Reichert, W.L., Laetz, C.A., Buzitis, J., Olson, O.P., Sol, S., and Collier, T.K. 2008. Improved flatfish health following remediation of a PAH-contaminated site in Eagle Harbor, Washington. Aquatic Toxicology 88: 277-288.
Myers, M.S., Landahl, J.T., Krahn, M.M., Johnson, L.L., and McCain, B.B. 1990. Overview of studies on liver carcinogenesis in English sole from Puget Sound; Evidence for a xenobiotic chemical etiology I: Pathology and epizootiology. Science of the Total Environment 94: 33-50.
Myers, M.S., Landahl, J.T., Krahn, M.M., and McCain, B.B. 1991. Relationships between hepatic neoplasms and related lesions and exposure to toxic chemicals in marine fish from the U.S. West Coastal. Environmental Health Perspectives 90: 7-15.
O'Connor, T.P. 2002. National distribution of chemical concentrations in mussels and oysters in the USA. Marine Environmental Research 53(2): 117-143.
O'Neill, S.M., and West, J.E. 2009. Marine Distribution, Life History Traits, and the Accumulation of Polychlorinated Biphenyls in Chinook Salmon from Puget Sound, Washington. Transactions of the American Fisheries Society 138(3): 616-632.
Partridge, V., Weakland, S., Long, E., Welch, K., Dutch, M., and Jones, M. 2009. Urban Waters Initiative, 2007; Sediment Quality in Elliott Bay. Washington Department of Ecology. Olympia, WA
Peterson, C.H., Rice, S.D., Short, J.W., Esler, D., Bodkin, J.L., Ballachey, B.E., and Irons, D.B. 2003. Long-Term Ecosystem Response to the Exxon Valdez Oil Spill. Science 302(5653): 2082-2086.
Puget Sound Action Team. 2007. Puget Sound Update: Ninth Report of the Puget Sound Assessment and Monitoring Program. Seattle, WA
Puget Sound Estuary Program. 1987. Recommended protocols for fish patholgy studies in Puget Sound. In Recommended Protocols and Guidelines for Measuring Selected Environmental Variables in Puget Sound. Prepaired by Tetra Tech, Inc., Bellevue, WA for U.S. Environmental Protection Agency, Region 10, Seattle, WA. (Looseleaf).
Puget Sound Estuary Program. 1988. Elliott Bay Action Program: Analysis of Toxic Problem Areas prepared for US Environmental Protection Agency, Region X, Seattle, WA.
Puget Sound Estuary Program. 1989a (revised). Recommended guidelines for measuring organic compounds in Puget Sound sediment and tissue samples. In Recommended Protocols and Guidlines for Measuring Selected Environmental Variables in Puget Sound. Prepared by PTI Environmental Services, Bellevue, WA. for U.S. Environmental Protection Agency, Region 10, Seattle, WA (Looseleaf).
Puget Sound Estuary Program. 1989b (Revised). Recommeded guidelines for measuring inorganic compounds in Puget Sound sediment and tissue samples. In Recommended Protocols and Guidelines for Measuring Selected Environmental Variables in Puget Sound. Prepaired by PTI Environmental Services, Bellevue, WA. (Looseleaf) for U.S. Environmental Protection Agency, Region 10, Seattle, WA.
Puget Sound Estuary Program. 1990. Recommended guidelines for sampling demersal fish. In Recommended Protocols and Guidelines for Measuring Selected Environmental Variables in Puget Sound. Prepaired by PTI Environmental Services, Bellevue, WA. for
U.S. Environmental Protection Agency, Region 10, Seattle, WA (Looseleaf).
Puget Sound Estuary Program. 1996a. Recommended guidelines for measuring metals in Puget Sound marine water, sediment and tissue samples, Prepared for the U.S. Environmental Protection Agency, Region 10, Seattle, WA, and Puget Sound Water Quality Authority by King County Environmental Lab, Seattle, WA, Seattle, WA. (Looseleaf).
Puget Sound Estuary Program. 1996b. Recommended guidelines for measuring organic compounds in Puget Sound marine water, sediment and tissue samples, Prepared for the U.S. Environmental Protection Agency, Region 10, Seattle, WA, and Puget Sound Water Quality Authority by King County Environmental Lab, Seattle, WA, Seattle, WA. (Looseleaf).
Ross, P.S., Ellis, G.M., Ikonomou, M.G., Barrett-Lennard, L.G., and Addison, R.F. 2000. High PCB concentrations in free-ranging Pacific killer whales, Orcinus orca: effects of age, sex and dietary preference. Marine Pollution Bulletin 40: 504-515.
Ross, P.S., Jeffries, S.J., M.B.Yunker, Addison, R.F., Ikonomou, M.G., and Calambokidis, J.C. 2004. Harbor seals (Phoca vitulina) in British Columbia, Canada, and Washington State, USA reveal a combination of local and global polychlorinated biphenyl, dioxin, and furan signals. Environmental Toxicology and Chemistry 23(1): 157-165.
Ross, P.S. 2006. Fireproof killer whales (Orcinus orca): flame retardant chemicals and the conservation imperative in the charismatic icon of British Columbia, Canada. Canadian Journal of Fisheries and Aquatic Science 63: 224-234.
Shen, G.T. 1995. Panel Findings and Recommendations: Based on the Final Comprehensive Review of the Puget Sound Ambient Monitoring Program, Puget Sound Water Quality Authority, P.O. Box 40900, Olympia, WA 98504-0900, Olympia, WA.
Sloan, J., and Gries, T. 2008. Dioxins, furans, and other contaminants in surface sediment and English sole collected from greater Elliott Bay (Seattle). Washington Department of Ecology. Olympia, WA
Spromberg, J.A., John, B.M., and Landis, W.G. 1998. Metapopulation dynamics: Indirect effects and multiple distinct outcomes in ecological risk assessment. Environmental Toxicology and Chemistry 17(8): 1640-1649.
Vines, C.A., Robbins, T., Griffin, F.J., and Cherr, G.N. 2000. The effects of diffusible creosote-derived compounds on development in Pacific herring (Clupea pallasi). Aquatic Toxicology 51(2): 225-239.
Weisbrod, A.V., Burkhard, L.P., Arnot, J., Mekenyan, O., Howard, P.H., Russom, C., Boethling, R., Sakuratani, Y., Traas, T., Bridges, T., Lutz, C., Bonnell, M., Woodburn, K., and Parkerton, T. 2007. Workgroup Report: Review of Fish Bioaccumulation Databases Used to Identify Persistent, Bioaccumulative, Toxic Substances. Environmental Health Perspectives 115(2): 255-261.
West, J.E., O'Neill, S.M., Lippert, G.R., and Quinnell, S.R. 2001. Toxic contaminants in marine and anadromous fish from Puget Sound, Washington: Results from the Puget Sound Ambient Monitoring Program Fish Component, 1989-1999. Technical Report. FTP01-14, Washington Department of Fish and Wildlife, Olympia, WA.
West, J.E., O'Neill, S.M., and Ylitalo, G.M. 2008. Spatial extent, magnitude, and patterns of persistent organochlorine pollutants in Pacific herring (Clupea pallasi) populations in the Puget Sound (USA) and Strait of Georgia (Canada). Science of the Total Environment 394(2-3): 369-378.
Wolfe, M.F., Schwartz, G.J.B., Singaram, S., Mielbrecht, E.E., Tjeerdema, R.S., and Sowby, M.L. 2001. Influence of dispersants on the bioavailability and trophic transfer of petroleum hydrocarbons to larval topsmelt (Atherinops affinis). Aquatic Toxicology 52(1): 49-60.