Even before the automobile was invented in the late 1800s, a self-made chemist, Charles Goodyear, paved the way for the modern tire by adding chemicals to alter the properties of natural rubber from the rubber tree. Natural rubber becomes soft and sticky in the heat of the summer, yet stiff and brittle in the cold of winter. In 1839, Goodyear added sulfur and heated the gummy material to produce a flexible yet durable form of rubber.
During World War II, supplies of natural rubber from Asia became difficult to come by, so chemical engineers in the U.S. and Russia came up with synthetic rubber made from crude oil. Their products fulfilled wartime needs for rubber until rubber-tree supplies resumed after the war. During the 1950s, chemists improved the formulas for synthetic rubber, and by the mid-1960s synthetic materials outpaced natural rubber in tires.
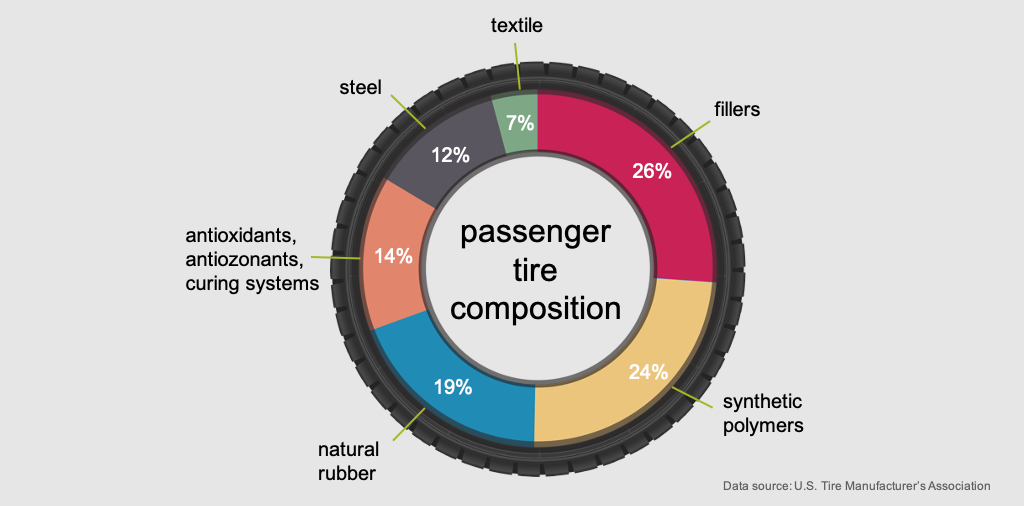
Today’s tubeless tires are a composite of up to 200 different chemicals. Each part of the tire — casing, sidewalls and tread — is manufactured to specific performance criteria, using a variety of natural and synthetic rubber compounds. Racing tires are very different from passenger tires, which are very different from truck tires. Various chemical formulations allow for tradeoffs among tire properties, such as resistance to aging, heat and wear, as well as the critical aspect of traction.